For example, the chlorine atoms in the 1 and 2 positions on the ring could both be pointing up above the ring (or down below it) or one could be pointing up and the other one down. It isn't too difficult to relate this to the ring case. Note: Geometric isomerism is explored on another page, although only with regard to carbon-carbon double bonds. This is one of the "chlorinated hydrocarbons" which caused so much environmental harm. One of these isomers was once commonly used as an insecticide known variously as BHC, HCH and Gammexane. Although there aren't any carbon-carbon double bonds, the bonds are still "locked" and unable to rotate. The chlorines and hydrogens can stick up and down at random above and below the ring and this leads to a number of geometric isomers. The ring delocalisation is permanently broken and a chlorine or bromine atom adds on to each carbon atom.įor example, if you bubble chlorine gas through hot benzene exposed to UV light for an hour, you get 1,2,3,4,5,6-hexachlorocyclohexane. In the presence of ultraviolet light (but without a catalyst present), hot benzene will also undergo an addition reaction with chlorine or bromine.
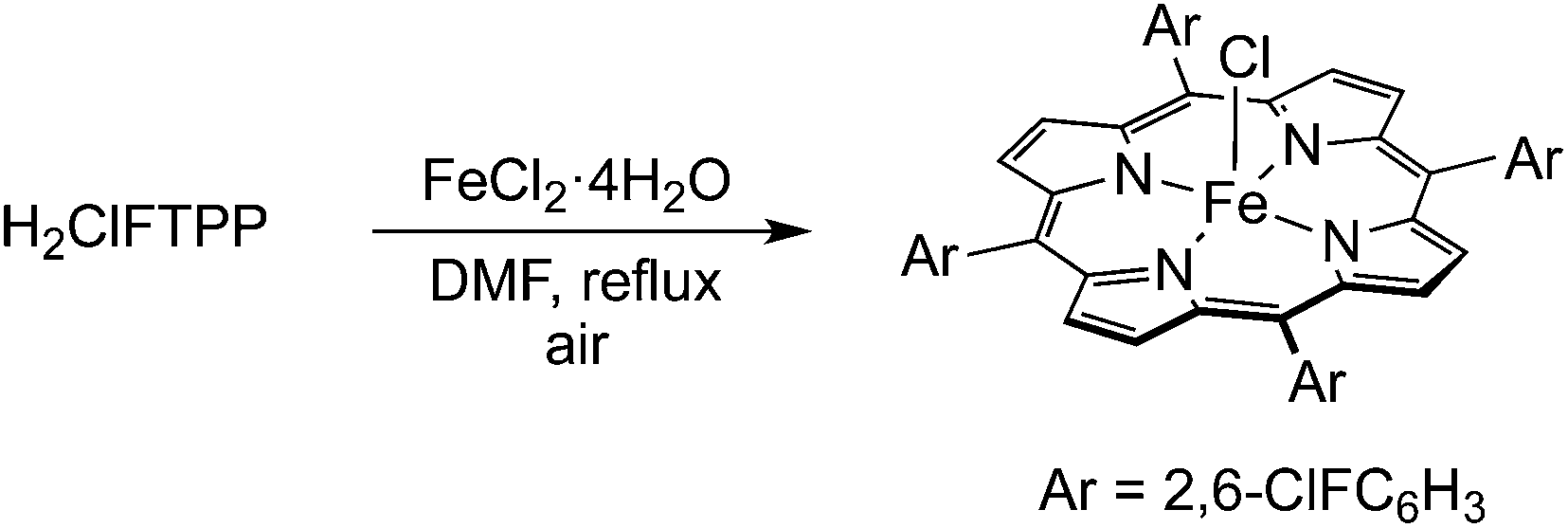
Use the BACK button on your browser to return to this page later. Note: Follow this link if you want the mechanism for the halogenation of benzene. Iron is usually used because it is cheaper and more readily available. The reaction between benzene and bromine in the presence of either aluminium bromide or iron gives bromobenzene.

The reaction between benzene and chlorine in the presence of either aluminium chloride or iron gives chlorobenzene. These compounds act as the catalyst and behave exactly like aluminium chloride, AlCl 3, or aluminium bromide, AlBr 3, in these reactions. It reacts with some of the chlorine or bromine to form iron(III) chloride, FeCl 3, or iron(III) bromide, FeBr 3. Strictly speaking iron isn't a catalyst, because it gets permanently changed during the reaction. The catalyst is either aluminium chloride (or aluminium bromide if you are reacting benzene with bromine) or iron. The reactions happen at room temperature. The mechanisms for several of these reactions are covered elsewhere on the site and you will find links to these other pages.īenzene reacts with chlorine or bromine in the presence of a catalyst, replacing one of the hydrogen atoms on the ring by a chlorine or bromine atom.
This page looks at the reactions of benzene and methylbenzene (toluene) with chlorine and bromine under various conditions. HALOGENATION OF BENZENE AND METHYLBENZENE Halogenation of benzene and methylbenzene

